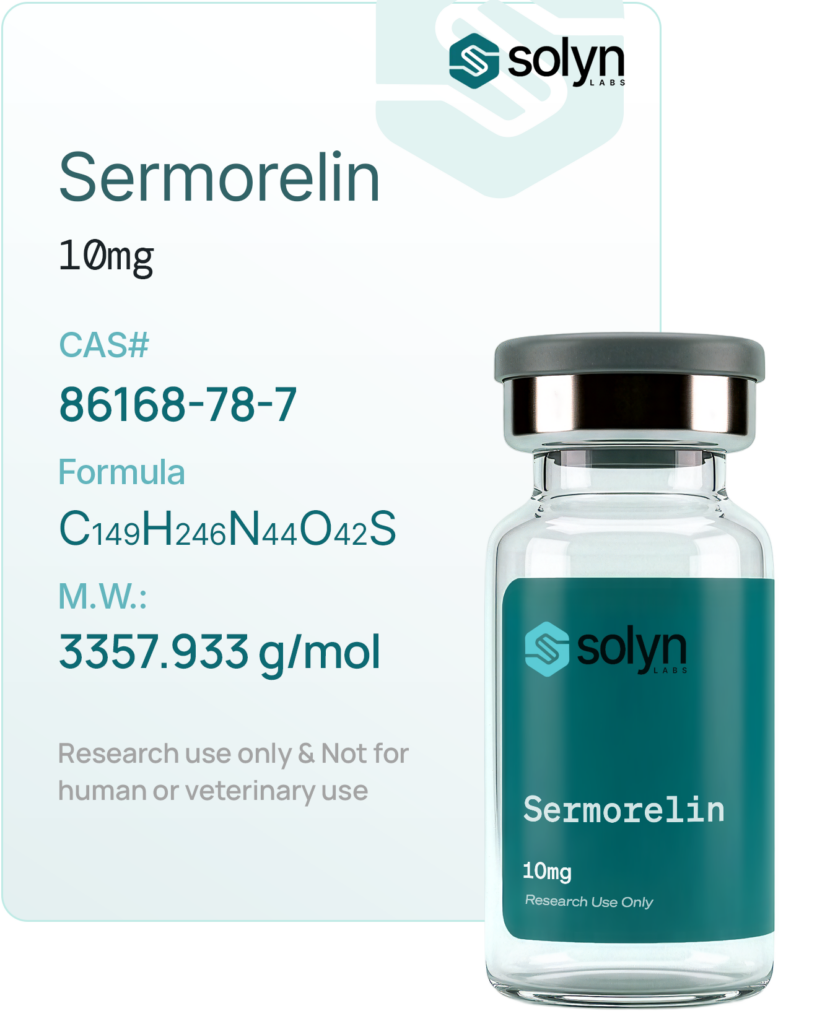
Order now &
this ships today
GLOW Stack
In stock
STATUS In Stock
batch number 1224
storage -20°C or below
Research Summary
Quantity & Testing
Storage & Handling
Quality Assurance Standards
Every batch undergoes comprehensive analytical testing to ensure pharmaceutical-grade quality. Complete analytical data and methodology details are included in the Certificate of Analysis (COA) provided with each order.
Storage & Handling
Store peptides in a cool, dry place away from light and moisture. For long-term stability, keep refrigerated at 2–8 °C. Avoid repeated freeze–thaw cycles. Handle using clean, dry tools and follow standard laboratory safety practices.
Purity Verification
High-Performance Liquid Chromatography (HPLC) with full chromatogram disclosure. All lots meet ≥99% purity specifications.
Structural Confirmation
Mass spectrometry (LC-MS) verifies molecular weight and structural integrity of each compound.
Composition Analysis
Analytical testing confirms accurate peptide content per vial and batch consistency.
Contamination Testing
Each lot undergoes testing for: Bacterial Endotoxin, Heavy metals, Bicrobial contamination and residual solvents (where applicable)
Related Research Compounds
Here goes your text ... Select any part of your text to access the formatting toolbar.